Investigation of the Reaction of HCl and Ice- Introduction
Marcia Joyce Isakson, Masters Thesis, Unversity of Texas 1994
(More information about the image at the right can be found at
Chester's home page)
 The atmospheric ozone layer has long protected humanity from the damaging
effects of solar radiation by absorbing harmful ultraviolet radiation.
However, atmospheric scientists have recently noticed a
dramatic decrease in ozone levels in the stratosphere. In January of 1993,
researchers discovered that ozone levels above the northern midlatitudes
were 13 to 14 percent below normal (Zurer, Pamela. (May, 1993). Chemical and
Engineering News, 8-18).. The
National Aeronautics and Space Administration
(NASA) documented that in 1993 the levels of ozone were the lowest in
fourteen years (Zurer 1993).
Many people blame the eruption of
Mount Pinatubo in the Philippines for the decrease, yet atmospheric
scientists claim that chlorofluorocarbons (CFC's) are the real culprits.
The atmospheric ozone layer has long protected humanity from the damaging
effects of solar radiation by absorbing harmful ultraviolet radiation.
However, atmospheric scientists have recently noticed a
dramatic decrease in ozone levels in the stratosphere. In January of 1993,
researchers discovered that ozone levels above the northern midlatitudes
were 13 to 14 percent below normal (Zurer, Pamela. (May, 1993). Chemical and
Engineering News, 8-18).. The
National Aeronautics and Space Administration
(NASA) documented that in 1993 the levels of ozone were the lowest in
fourteen years (Zurer 1993).
Many people blame the eruption of
Mount Pinatubo in the Philippines for the decrease, yet atmospheric
scientists claim that chlorofluorocarbons (CFC's) are the real culprits.
Since chlorofluorocarbons are used in industry as refrigerants and
are sold to the public in products
such as aerosol cans, the theories that have been
proposed have come under considerable scrutiny. There is a need
to investigate these theories in complete detail,
so that a solution that is
scientifically valid may be formulated.
Theories advanced by atmospheric scientists rely on many
reactions taking place in order for the chlorofluorocarbons to destroy the
ozone layer. This work investigated the role of heterogeneous reactions
in the destruction of ozone in the stratosphere.
Specifically, we investigate
the reaction of hydrogen chloride (HCl) and solid water
(ice).
Normally ozone is continually created and destroyed via the following
reactions:
O2 + hv --> O + O
O + O2 --> O3
O3 + hv --> O2 + O
These reactions take place about 40 kilometers above the earth in the
stratosphere.
It is theorized that the CFC's rise into
the upper atmosphere where they are photolyzed to produce another CFC and
a chlorine free radical (Zurer 1993). One such reaction is:
CCl2F2 (CFC-12) + hv --> CClF2 + Cl
Free chlorine radicals act as catalysts which deplete the population of
O3 molecules available for the reaction 1.3 by the process:
Cl + O3 --> ClO + O2
ClO + O --> Cl + O2
O3 + O --> 2O2 (net)
Because the chlorine radical is regenerated in this process, it
is available to destroy many other O3 molecules by the same process.
One cycle that atmospheric scientists are particularly interested in
understanding is the seasonal
ozone depletion above the Antarctic. The Antarctic is an
interesting system to study because it has suffered extensive damage,
i.e. the well publicized ozone ``hole''.
Additionally, in the winter, it becomes a closed system.
During this season,
the winds begin to circle above the polar region,
creating what is called a polar vortex. The winds of the polar
vortex region do not mix with the rest of the atmosphere until
late spring.
The mechanism described in equation 1.5
does not explain the
destruction of the ozone layer above the Antarctic for two reasons.
First, it does not provide for a seasonal depletion of ozone
as is observed above the Antarctic. Secondly, there are not enough
O atoms present
in the altitudes in which the most prominent destruction has occurred.
In fact, these reactions have been shown to be insignificant at
40 km, stratospheric altitudes (Zurer 1993). Therefore another theory
has to be considered.
The theory that accounts for seasonal depletion of ozone
takes into account the reservoir species.
These species are nodes in holding reactions. These reactions
compete with ozone destroying mechanisms and have the effect of
removing destructive species from the ozone cycle Wayne, Chemistry of Atmospheres
1991).The most prominent of these reservoir species is hydrogen chloride,
HCl, which accounts
for about 70 percent of atmospheric Cl.
To introduce a seasonal cycle,
a mechanism is needed to explain how these reservoir species are converted
into species active in for ozone destruction.
It is proposed that HCl and ClONO2 react on the surface of
polar stratospheric
ice particles in the clouds
and produce molecular chlorine (Cl2) and
hypochlorous acid (HOCl). Tolbert, Science 240, 1988).
The clouds in the
polar vortex region, called polar stratospheric clouds (PSC's),
carry ice crystals which provide surfaces on which
the above reactions can take place.
There are two types of cloud particles
present: PSC Type (I), smaller particles (1 $\mu$m)
consisting of nitric acid
trihydride (NAT), and PSC Type (II), larger particles (10 $\mu$m)
constisting of
mostly frozen water condensed on a NAT nucleus
Fahey, J. Geo. Res., 94 1989). The reservoir species react on the
surfaces of these PSC's all winter long, creating Cl2 and HOCl. When
the sun returns in the spring, the Cl2 and HOCl
are photolyzed, creating a deluge
of ozone destroying radicals. Thus, the ozone hole is created over the
Antarctic in the spring.
Experiment
Rudye McGlothlin is currently working on this project for his Senior Thesis.
 In this photo the top hat piece which holds the Silicon surface is being heated to 1000C to clean the Silicon. Once the surface is clean it will be cooled down with liquid nitrogen and dosed with water to form an ice surface.
In this photo the top hat piece which holds the Silicon surface is being heated to 1000C to clean the Silicon. Once the surface is clean it will be cooled down with liquid nitrogen and dosed with water to form an ice surface.
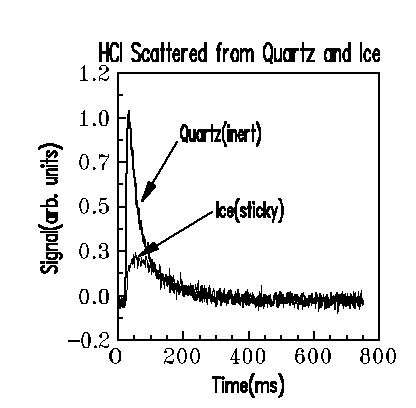 Recent results on measuring the sticking coefficient of HCl on Ice.
Recent results on measuring the sticking coefficient of HCl on Ice.
 The atmospheric ozone layer has long protected humanity from the damaging
effects of solar radiation by absorbing harmful ultraviolet radiation.
However, atmospheric scientists have recently noticed a
dramatic decrease in ozone levels in the stratosphere. In January of 1993,
researchers discovered that ozone levels above the northern midlatitudes
were 13 to 14 percent below normal (Zurer, Pamela. (May, 1993). Chemical and
Engineering News, 8-18).. The
National Aeronautics and Space Administration
(NASA) documented that in 1993 the levels of ozone were the lowest in
fourteen years (Zurer 1993).
Many people blame the eruption of
Mount Pinatubo in the Philippines for the decrease, yet atmospheric
scientists claim that chlorofluorocarbons (CFC's) are the real culprits.
The atmospheric ozone layer has long protected humanity from the damaging
effects of solar radiation by absorbing harmful ultraviolet radiation.
However, atmospheric scientists have recently noticed a
dramatic decrease in ozone levels in the stratosphere. In January of 1993,
researchers discovered that ozone levels above the northern midlatitudes
were 13 to 14 percent below normal (Zurer, Pamela. (May, 1993). Chemical and
Engineering News, 8-18).. The
National Aeronautics and Space Administration
(NASA) documented that in 1993 the levels of ozone were the lowest in
fourteen years (Zurer 1993).
Many people blame the eruption of
Mount Pinatubo in the Philippines for the decrease, yet atmospheric
scientists claim that chlorofluorocarbons (CFC's) are the real culprits. In this photo the top hat piece which holds the Silicon surface is being heated to 1000C to clean the Silicon. Once the surface is clean it will be cooled down with liquid nitrogen and dosed with water to form an ice surface.
In this photo the top hat piece which holds the Silicon surface is being heated to 1000C to clean the Silicon. Once the surface is clean it will be cooled down with liquid nitrogen and dosed with water to form an ice surface. Recent results on measuring the sticking coefficient of HCl on Ice.
Recent results on measuring the sticking coefficient of HCl on Ice.